Do you shy away from buying Radium dialed & handed watches?
Flatfoot
·No, I don’t shy away from them.
However I spent some time evaluating radium lume before adding pieces to my collection. My opinion to proceed was based on what I was able to find at the time, and that data satisfied my personal risk/reward balance.
I believe the highest risk for my own personal assessment would be the ingestion of any flaking radium lume. I manage that by not proceeding beyond opening a caseback for taking a photo or making a simple regulation adjustment.
A second risk is the generation of radon gas. Without getting into specifics, any radon gas that may be generated would be dissipated rather well by how I store my watches.
So that leaves the risk of any radiation exposure from the watch itself while wearing it - both dial side and caseback side.
Taking the following watches to my watchmaker for a Geiger counter assessment shows truly minimal transfer through the caseback into my wrist. However the dial side may be quite hot. So yes, I don’t sleep with any watch on anyway- and specifically will not sleep with a radium dialed watch on my wrist and tucked under my head as I sleep.
And any watch service which may be done by my guys is then made with the knowledge that there is radium on the dial.
With the risks managed to suit me as described above, I then find the aesthetics too good to pass up. So I wear them in rotation and enjoy them.
However I spent some time evaluating radium lume before adding pieces to my collection. My opinion to proceed was based on what I was able to find at the time, and that data satisfied my personal risk/reward balance.
I believe the highest risk for my own personal assessment would be the ingestion of any flaking radium lume. I manage that by not proceeding beyond opening a caseback for taking a photo or making a simple regulation adjustment.
A second risk is the generation of radon gas. Without getting into specifics, any radon gas that may be generated would be dissipated rather well by how I store my watches.
So that leaves the risk of any radiation exposure from the watch itself while wearing it - both dial side and caseback side.
Taking the following watches to my watchmaker for a Geiger counter assessment shows truly minimal transfer through the caseback into my wrist. However the dial side may be quite hot. So yes, I don’t sleep with any watch on anyway- and specifically will not sleep with a radium dialed watch on my wrist and tucked under my head as I sleep.
And any watch service which may be done by my guys is then made with the knowledge that there is radium on the dial.
With the risks managed to suit me as described above, I then find the aesthetics too good to pass up. So I wear them in rotation and enjoy them.
Tet
·I'm happy enough wearing watches with radium, not pocket watches though, don't want radium so close to the old crown jewels.
Dsloan
·I've actively avoided buying a few pieces because of radium lume. I have a toddler and am paranoid about him somehow breaking a watch and touching/ingesting the lume.
Also, I'm glad that people are mentioning the radon issue. That seems like potentially a bigger concern than the alpha particles from the radium lume itself.
Also, I'm glad that people are mentioning the radon issue. That seems like potentially a bigger concern than the alpha particles from the radium lume itself.
Texas Toast
·No claim of authority, and might be a minor issue, but I have a hard time dismissing the radon aspect.
As part of a good thread on the topic https://omegaforums.net/threads/radiation-concerns-part-2.79191/page-4#post-1307034 I measured somewhat disconcerting radon levels in open air near an old Navy Deck clock on the mantle.
Radon levels dropped significantly once away from the clock. The amount of lume on the clock is large compared to a single watch (though a radium watch collection might be equivalent). Storage method surely makes a big difference on exposure. Exposure dynamics are certainly different between a home mantle clock and a wrist-worn watch.
Still, my guess is less radon is better than more radon so I've moved the clock to the garage for now. Ditto a WWII Omega pocket watch with big radium-coated hands, numbers, and hour markers.
As part of a good thread on the topic https://omegaforums.net/threads/radiation-concerns-part-2.79191/page-4#post-1307034 I measured somewhat disconcerting radon levels in open air near an old Navy Deck clock on the mantle.
Radon levels dropped significantly once away from the clock. The amount of lume on the clock is large compared to a single watch (though a radium watch collection might be equivalent). Storage method surely makes a big difference on exposure. Exposure dynamics are certainly different between a home mantle clock and a wrist-worn watch.
Still, my guess is less radon is better than more radon so I've moved the clock to the garage for now. Ditto a WWII Omega pocket watch with big radium-coated hands, numbers, and hour markers.
Edited:
timecube
·I've actively avoided buying a few pieces because of radium lume. I have a toddler and am paranoid about him somehow breaking a watch and touching/ingesting the lume.
Also, I'm glad that people are mentioning the radon issue. That seems like potentially a bigger concern than the alpha particles from the radium lume itself.
__________
Update:
it seems like non-volatile daughter products of Ra-226 sitting inside the case are the source of most of the gammas. I still think the specific risk from Rn-222 being released wouldn't be a big deal.
Some interesting notes from the NRC people:
https://www.nrc.gov/docs/ML0731/ML073120009.pdf
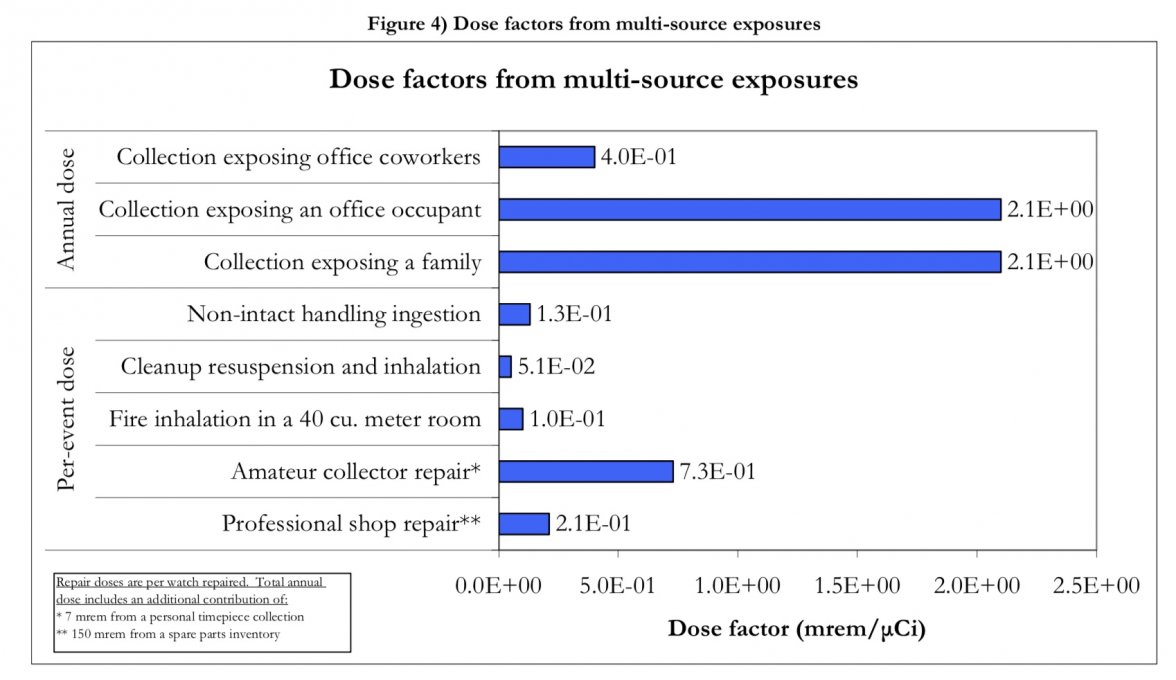
Toddler-watch-breaking-and-lume-ingestion seems to be a relatively low risk factor, compared with exposing your unshielded dial to the toddler on a routine basis.
And apparently, repairs by amateur "enthusiasts" are considered most risky of all!
DaveK
·seekingseaquest
·Braindrain
·So....
what numbers do you see through the caseback?
I can't remember but it was very low. However, as others have mentioned, it's not the radiation that's the concern, more the radon that is released. However, without summarizing the whole report from a few yrs back, the readings were hardly completed in a controlled environment.
DaveK
·Radium, no T next to Swiss and color indicates radium to me.
asrnj77
·Thank you. I thought there was a time early in the beginnings of tritium’s useage before they realized that differentiating with a T would be helpful.
The earliest I’ve seen anybody talk about Omega using tritium was 1962. I think Rolex started in 1963. If you’re confident that yours is a ‘61 then I’d lean with radium but a Geiger counter could solve the mystery quickly
Helvetia History
·No claim of authority, and might be a minor issue, but I have a hard time dismissing the radon aspect.
As part of a good thread on the topic https://omegaforums.net/threads/radiation-concerns-part-2.79191/page-4#post-1307034 I measured somewhat disconcerting radon levels in open air near an old Navy Deck clock on the mantle.
Radon levels dropped significantly once away from the clock. The amount of lume on the clock is large compared to a single watch (though a radium watch collection might be equivalent). Storage method surely makes a big difference on exposure. Exposure dynamics are certainly different between a home mantle clock and a wrist-worn watch.
Still, my guess is less radon is better than more radon so I've moved the clock to the garage for now. Ditto a WWII Omega pocket watch with big radium-coated hands, numbers, and hour markers.
Hi,
I found out recently that almost all of the Radon gas detectors available don't measure Radon directly but actually measure Alpha particles and then, as there should be no other source of Alpha radiation in the standard home, assume this is from radon gas and extrapolate the Radon reading from this.
If you put the meter by a Radium watch or clock it is obviously going to to read the Alpha particles from the Radium and wrongly assume this is from Radon gas. This means that high readings of Radon you are seeing next to your clock are probably directly from the Radium on the dial and not just Radon gas.
In the circumstances these detectors are sold for normally this is fine as any alpha particles are not likely to be from any other source in the home but if you want to measure vintage Radium dialled watches I think they are pretty useless. You are putting them next to an Alpha particle source and expecting to get a reading from the gas the dial gives off not from the Radium itself but the meter cannot differentiate.
I think a lot of people are being misled by their Radon gas detectors as they are naturally assuming that the reader is measuring the gas directly when it isn't.
Here is a thread: https://www.mwrforum.net/forums/showthread.php?87886-Any-chemists-out-there
Thanks. Carl.
Texas Toast
·Thank you, Carl. I hadn't considered the potentially confounding effect of nearby radium.
You are correct on alpha particle detection and this was mentioned by @padders in the earlier 'radiation part 2' thread. In the same thread @Dan S points out the typically isolated nature or radon-source alpha emissions inside a shielded sample chamber (Dan also contributed to the MWR thread you reference)
Airthing's website explains the basics of meter operation:
https://www.airthings.com/resources/radon-detector
A photodetector senses alpha decay in a chamber as a proxy for Radon and "This chamber is where the detector analyzes your home’s air. Chrome is used in the chamber, which is constructed in such a way that it is a sort of cage against which unwanted particles are barred".
While they do not specifically say, I would think the chamber blocks external (radium dial) alpha emissions.
From the NRC wording it wouldn't seem to take much to effectively isolate alpha emissions in a chamber;
https://www.nrc.gov/about-nrc/radiation/health-effects/radiation-basics.html#alpha
"In general, alpha particles have a very limited ability to penetrate other materials. In other words, these particles of ionizing radiation can be blocked by a sheet of paper, skin, or even a few inches of air."
Could the 0.2mm chamber orifice mentioned in the Airthings writeup be a sneak path (break in the alpha shield) and was I close enough that alpha particles were still reaching the meter (about 2-3" away from clock)? Given NRC wording, wouldn't a clock/watch case be a decent alpha shield anyway?
Bad data makes for bad decisions, and I'd love to be wrong about what my meter is telling me. For now I'm inclined to believe the readings until it is clear that 1) alpha particles from dial radium would reach the meter and 2) the meter chamber is 'leaky' and measuring these external alpha particles.
I may be over-reacting even if data is correct but that's a separate matter related to my not understanding what constitutes acceptable exposure.
You are correct on alpha particle detection and this was mentioned by @padders in the earlier 'radiation part 2' thread. In the same thread @Dan S points out the typically isolated nature or radon-source alpha emissions inside a shielded sample chamber (Dan also contributed to the MWR thread you reference)
Airthing's website explains the basics of meter operation:
https://www.airthings.com/resources/radon-detector
A photodetector senses alpha decay in a chamber as a proxy for Radon and "This chamber is where the detector analyzes your home’s air. Chrome is used in the chamber, which is constructed in such a way that it is a sort of cage against which unwanted particles are barred".
While they do not specifically say, I would think the chamber blocks external (radium dial) alpha emissions.
From the NRC wording it wouldn't seem to take much to effectively isolate alpha emissions in a chamber;
https://www.nrc.gov/about-nrc/radiation/health-effects/radiation-basics.html#alpha
"In general, alpha particles have a very limited ability to penetrate other materials. In other words, these particles of ionizing radiation can be blocked by a sheet of paper, skin, or even a few inches of air."
Could the 0.2mm chamber orifice mentioned in the Airthings writeup be a sneak path (break in the alpha shield) and was I close enough that alpha particles were still reaching the meter (about 2-3" away from clock)? Given NRC wording, wouldn't a clock/watch case be a decent alpha shield anyway?
Bad data makes for bad decisions, and I'd love to be wrong about what my meter is telling me. For now I'm inclined to believe the readings until it is clear that 1) alpha particles from dial radium would reach the meter and 2) the meter chamber is 'leaky' and measuring these external alpha particles.
I may be over-reacting even if data is correct but that's a separate matter related to my not understanding what constitutes acceptable exposure.
Koelsh
·Helvetia History
·Thank you, Carl. I hadn't considered the potentially confounding effect of nearby radium.
You are correct on alpha particle detection and this was mentioned by @padders in the earlier 'radiation part 2' thread. In the same thread @Dan S points out the typically isolated nature or radon-source alpha emissions inside a shielded sample chamber (Dan also contributed to the MWR thread you reference)
Airthing's website explains the basics of meter operation:
https://www.airthings.com/resources/radon-detector
A photodetector senses alpha decay in a chamber as a proxy for Radon and "This chamber is where the detector analyzes your home’s air. Chrome is used in the chamber, which is constructed in such a way that it is a sort of cage against which unwanted particles are barred".
While they do not specifically say, I would think the chamber blocks external (radium dial) alpha emissions.
From the NRC wording it wouldn't seem to take much to effectively isolate alpha emissions in a chamber;
https://www.nrc.gov/about-nrc/radiation/health-effects/radiation-basics.html#alpha
"In general, alpha particles have a very limited ability to penetrate other materials. In other words, these particles of ionizing radiation can be blocked by a sheet of paper, skin, or even a few inches of air."
Could the 0.2mm chamber orifice mentioned in the Airthings writeup be a sneak path (break in the alpha shield) and was I close enough that alpha particles were still reaching the meter (about 2-3" away from clock)? Given NRC wording, wouldn't a clock/watch case be a decent alpha shield anyway?
Bad data makes for bad decisions, and I'd love to be wrong about what my meter is telling me. For now I'm inclined to believe the readings until it is clear that 1) alpha particles from dial radium would reach the meter and 2) the meter chamber is 'leaky' and measuring these external alpha particles.
I may be over-reacting even if data is correct but that's a separate matter related to my not understanding what constitutes acceptable exposure.
Thanks. I had read the article on the way the meters work and had taken the part about keeping out unwanted particles to mean particles other than Alpha radiation that might confuse the reading. I agree they could mean it keeps out stray Alpha particles, isolating the air in the chamber and then just reading the Alpha radiation given of by Radon in that trapped air and that would make for a more accurate reading and makes sense.
I am aware that Alpha particles don't travel far, however that does seem to tie in with you getting high readings when within a couple of inches of your clock.
I only know what I have been told by other more knowledgeable than myself but just thought it useful for people to know what these meters are actually reading and that, not being designed to read Radon gas directly from a nearby Radium source but rather the amount in the ambient air in a house, that you might not be seeing what you think you are seeing with these readings. Of course it is best to err on the side of caution with these things and I'm sure Radon gas is a real concern and needs to be taken seriously.
Thanks. Carl.
timecube
·I just upgraded my dosimeter because I have a watch that got off the scale...
I was able to measure almost 90 uSv/h and over 13,000 CPM
What do you guys think? Would you wear this?
Wearing it dial-side down, 16 hours a day for a year would put you at the low end of the red section of this chart:
a slightly increased risk of cancer, but wearing such a beautiful watch should be worth it!
ᵐᵃʸᵇᵉ
Texas Toast
·Based on the radon measurement via alpha-detection as discussed up-thread I wanted to further understand whether radon measurements were influenced by proximal radium.
Two exercises were run in parallel.
1) Contact Airthings directly about the Home meter and our question about meter accuracy with radium nearby
2) Do a semi-sealed enclosure test with a pocket watch and Airthings meter having ~15-20cm separation (longer than alpha particle travel distances as I understand it)
Short version of results: a) The meter is only reading the radon levels (via alpha particle proxy) and not being fooled by any radium in the neighborhood b) one can achieve quite a high concentration of radon with a single radium-rich dial in a small closed volume.
Just reporting some findings. Still pondering what they mean and any other thoughts are of interest. I tried to follow the radiation dosage chart above and it only confirmed that I don't understand much about relative radiation risks.
-----
Long version of results:
1) Company responds (nice!) that meter is accurately reading radon levels and not being influenced by proximal radium
Hi David,
My name is Laoise and I am the Customer Support Specialist here at Airthings. I asked our in-house CERN scientist, and this was his reply:
Your question: In other words, is the meter still accurate when in close proximity (say <10cm apart) to a strong alpha emitter like these radium-bearing timepieces?
The answer is yes. The alpha particles from the radium in the watches can not reach the photodiode sensor inside the diffusion (decay) chamber.
If there is anything else I can help you with, please let me know! Thanks for being an Airthings customer 😁
All the best,
Laoise
Airthings support
2) My testing would seem to confirm the answer from Airthings.
Watch and meter were sealed in a generic food storage container. Surely not hermetic, but decent, and separation should be enough to diminish radium alpha particle influence. Airthing meter spiked in a matter of hours to saturation at 500.0 pCi/L 😲.
For reference, my typical home levels are 0.5-1.5 pCi/L depending on the meter location and day. Readings typically take a day to begin. Problematic levels begin in the 4 pCi/L range (again as I understand it).
I removed meter to inside and put watch in garage. After a reset it again quickly spiked to 500 pCi/L, presumably from retained radon in the diffusion chamber. Then tried to "air out the chamber" by going outside on a breezy day and waving it around for 15 seconds. Reset. Still spiked to 500 pCi/L quickly, though not quite as quick. Reset, and just let sit for a while ... down to 194pCi/L after ~12 hrs. Reset. After 24 hours I'm quickly approaching a typical ~1pCi/L. Sounds a lot like captured radon in the chamber slowly dissipating/decaying, all without any radium nearby.
Two exercises were run in parallel.
1) Contact Airthings directly about the Home meter and our question about meter accuracy with radium nearby
2) Do a semi-sealed enclosure test with a pocket watch and Airthings meter having ~15-20cm separation (longer than alpha particle travel distances as I understand it)
Short version of results: a) The meter is only reading the radon levels (via alpha particle proxy) and not being fooled by any radium in the neighborhood b) one can achieve quite a high concentration of radon with a single radium-rich dial in a small closed volume.
Just reporting some findings. Still pondering what they mean and any other thoughts are of interest. I tried to follow the radiation dosage chart above and it only confirmed that I don't understand much about relative radiation risks.
-----
Long version of results:
1) Company responds (nice!) that meter is accurately reading radon levels and not being influenced by proximal radium
Hi David,
My name is Laoise and I am the Customer Support Specialist here at Airthings. I asked our in-house CERN scientist, and this was his reply:
Your question: In other words, is the meter still accurate when in close proximity (say <10cm apart) to a strong alpha emitter like these radium-bearing timepieces?
The answer is yes. The alpha particles from the radium in the watches can not reach the photodiode sensor inside the diffusion (decay) chamber.
If there is anything else I can help you with, please let me know! Thanks for being an Airthings customer 😁
All the best,
Laoise
Airthings support
2) My testing would seem to confirm the answer from Airthings.
Watch and meter were sealed in a generic food storage container. Surely not hermetic, but decent, and separation should be enough to diminish radium alpha particle influence. Airthing meter spiked in a matter of hours to saturation at 500.0 pCi/L 😲.
For reference, my typical home levels are 0.5-1.5 pCi/L depending on the meter location and day. Readings typically take a day to begin. Problematic levels begin in the 4 pCi/L range (again as I understand it).
I removed meter to inside and put watch in garage. After a reset it again quickly spiked to 500 pCi/L, presumably from retained radon in the diffusion chamber. Then tried to "air out the chamber" by going outside on a breezy day and waving it around for 15 seconds. Reset. Still spiked to 500 pCi/L quickly, though not quite as quick. Reset, and just let sit for a while ... down to 194pCi/L after ~12 hrs. Reset. After 24 hours I'm quickly approaching a typical ~1pCi/L. Sounds a lot like captured radon in the chamber slowly dissipating/decaying, all without any radium nearby.
Dan S
·Based on the radon measurement via alpha-detection as discussed up-thread I wanted to further understand whether radon measurements were influenced by proximal radium.
Two exercises were run in parallel.
1) Contact Airthings directly about the Home meter and our question about meter accuracy with radium nearby
2) Do a semi-sealed enclosure test with a pocket watch and Airthings meter having ~15-20cm separation (longer than alpha particle travel distances as I understand it)
Short version of results: a) The meter is only reading the radon levels (via alpha particle proxy) and not being fooled by any radium in the neighborhood b) one can achieve quite a high concentration of radon with a single radium-rich dial in a small closed volume.
Just reporting some findings. Still pondering what they mean and any other thoughts are of interest. I tried to follow the radiation dosage chart above and it only confirmed that I don't understand much about relative radiation risks.
-----
Long version of results:
1) Company responds (nice!) that meter is accurately reading radon levels and not being influenced by proximal radium
Hi David,
My name is Laoise and I am the Customer Support Specialist here at Airthings. I asked our in-house CERN scientist, and this was his reply:
Your question: In other words, is the meter still accurate when in close proximity (say <10cm apart) to a strong alpha emitter like these radium-bearing timepieces?
The answer is yes. The alpha particles from the radium in the watches can not reach the photodiode sensor inside the diffusion (decay) chamber.
If there is anything else I can help you with, please let me know! Thanks for being an Airthings customer 😁
All the best,
Laoise
Airthings support
2) My testing would seem to confirm the answer from Airthings.
Watch and meter were sealed in a generic food storage container. Surely not hermetic, but decent, and separation should be enough to diminish radium alpha particle influence. Airthing meter spiked in a matter of hours to saturation at 500.0 pCi/L 😲.
For reference, my typical home levels are 0.5-1.5 pCi/L depending on the meter location and day. Readings typically take a day to begin. Problematic levels begin in the 4 pCi/L range (again as I understand it).
I removed meter to inside and put watch in garage. After a reset it again quickly spiked to 500 pCi/L, presumably from retained radon in the diffusion chamber. Then tried to "air out the chamber" by going outside on a breezy day and waving it around for 15 seconds. Reset. Still spiked to 500 pCi/L quickly, though not quite as quick. Reset, and just let sit for a while ... down to 194pCi/L after ~12 hrs. Reset. After 24 hours I'm quickly approaching a typical ~1pCi/L. Sounds a lot like captured radon in the chamber slowly dissipating/decaying, all without any radium nearby.
This is all consistent with my observations and the information from the company sounds absolutely correct to me (i.e. the detector is shielded from ambient radiation). As you found, the radon emitted from a watch if contained in a small container will reach high concentration levels, and it doesn't dissipate readily without convection because radon is very dense. The only additional thing to keep in mind is that health effects are mainly associated with the total dose of radon inhaled, which is calculated as being due to a constant background level that is inhaled multiple times per minute, assuming a standard tidal volume. Despite the high concentration of radon in that container, the total dose of radon is tiny because of the small volume. To get a feeling for this dose issue, here is a rough calculation that I posted on WUS a while back, which was based on a measurement I made of 125 pCi/L when I put my meter in a container with 10 radium watches. My radium watches are not as heavily lumed as yours, and are more representative of 1950s wristwatches.
----------
For a back-of-the-envelope wildly conservative estimate of the maximum dose one could potentially receive from opening a watch box, let's compare the total daily dose of radon one receives at outdoors background levels (approximately one-tenth the level considered hazardous) to an exaggerated dose that one could possibly receive from two daily episodes of sticking one's head into a sealed container of watches and breathing deeply for 30 seconds each time (1 minute total).
Outdoor background: (0.4 pCi/L)*(20,000 inhalations/day)*(0.5 L/inhalation) = 4,000 pCi.
Sealed container: (125 pCi/L)*(15 inhalations/minute)*(0.5 L/inhalation) = 937 pCi.
So even in the unlikely case that you would breath deeply for a full minute from the air at the bottom of a closed container holding 10 radium-lumed watches (and assuming that there is enough radon-laden air in the watch box to fill your lungs for that whole time), your total dose would only be about one-quarter of the dose you receive daily from outdoors ambient levels, which is itself 10 times smaller than the dose considered to be dangerous. So this one-minute deep-breathing exercise from your watch box represents about 2% of a hazardous daily dose of radon. And of course, because the assumptions used in this calculation are exaggerated (e.g. there is not actually 7.5L of radon-laden air in the watch-box, it's not likely that you would inhale the air from the bottom of the box, etc.), the actual dose you would receive from opening your watch box would be much lower.
------------
To make the calculation more directly relevant to your experiment, you could replace the total dose from the sealed container to be the actual total amount of radon in that container, which is obviously the upper limit. This would be (500 pCi/L)*(1L)=500 pCi. This is assuming it was a 1L container. Of course it's unlikely you would be able to inhale all of the radon out of that container without trying really hard, but that is the maximum dose you could get, compared to about 4,000 pCi each day from outdoors background, which is the absolute minimum we are all exposed to.
These are all just numbers, and I'm not telling people whether or not to be concerned about increasing their exposure to radon by even a small amount. But data are data, and we can't ignore them. When you multiply the concentration by the volume (which is the relevant calculation), the numbers make more sense.
Edited:
timecube
·This is all consistent with my observations and the information from the company sounds absolutely correct to me (i.e. the detector is shielded from ambient radiation). As you found, the radon emitted from a watch if contained in a small container will reach high concentration levels, and it doesn't dissipate readily without convection because radon is very dense. The only additional thing to keep in mind is that health effects are mainly associated with the total dose of radon inhaled, which is calculated as being due to a constant background level that is inhaled multiple times per minute, assuming a standard tidal volume. Despite the high concentration of radon in that container, the total dose of radon is tiny because of the small volume. To get a feeling for this dose issue, here is a rough calculation that I posted on WUS a while back, which was based on a measurement I made of 125 pCi/L when I put my meter in a container with 10 radium watches. My radium watches are not as heavily lumed as yours, and are more representative of 1950s wristwatches.
----------
For a back-of-the-envelope wildly conservative estimate of the maximum dose one could potentially receive from opening a watch box, let's compare the total daily dose of radon one receives at outdoors background levels (approximately one-tenth the level considered hazardous) to an exaggerated dose that one could possibly receive from two daily episodes of sticking one's head into a sealed container of watches and breathing deeply for 30 seconds each time (1 minute total).
Outdoor background: (0.4 pCi/L)*(20,000 inhalations/day)*(0.5 L/inhalation) = 4,000 pCi.
Sealed container: (125 pCi/L)*(15 inhalations/minute)*(0.5 L/inhalation) = 937 pCi.
So even in the unlikely case that you would breath deeply for a full minute from the air at the bottom of a closed container holding 10 radium-lumed watches (and assuming that there is enough radon-laden air in the watch box to fill your lungs for that whole time), your total dose would only be about one-quarter of the dose you receive daily from outdoors ambient levels, which is itself 10 times smaller than the dose considered to be dangerous. So this one-minute deep-breathing exercise from your watch box represents about 2% of a hazardous daily dose of radon. And of course, because the assumptions used in this calculation are exaggerated (e.g. there is not actually 7.5L of radon-laden air in the watch-box, it's not likely that you would inhale the air from the bottom of the box, etc.), the actual dose you would receive from opening your watch box would be much lower.
------------
To make the calculation more directly relevant to your experiment, you could replace the total dose from the sealed container to be the actual total amount of radon in that container, which is obviously the upper limit. This would be (500 pCi/L)*(1L)=500 pCi. This is assuming it was a 1L container. Of course it's unlikely you would be able to inhale all of the radon out of that container without trying really hard, but that is the maximum dose you could get, compared to about 4,000 pCi each day from outdoors background, which is the absolute minimum we are all exposed to.
These are all just numbers, and I'm not telling people whether or not to be concerned about increasing their exposure to radon by even a small amount. But data are data, and we can't ignore them. When you multiply the concentration by the volume (which is the relevant calculation), the numbers make more sense.